Taste assessment is the earliest point at which palatability risk can be measured, understood, and controlled. Most formulation approaches attempt to correct taste late in development. By then, the feasible solution space is already constrained.
At Senopsys, we use GCP-compliant human taste panels and quantitative sensory methods to transform taste from a subjective observation into a measurable design parameter. This enables formulation strategies grounded in data—not trial and error.
FlavorMetrics℠ Profile
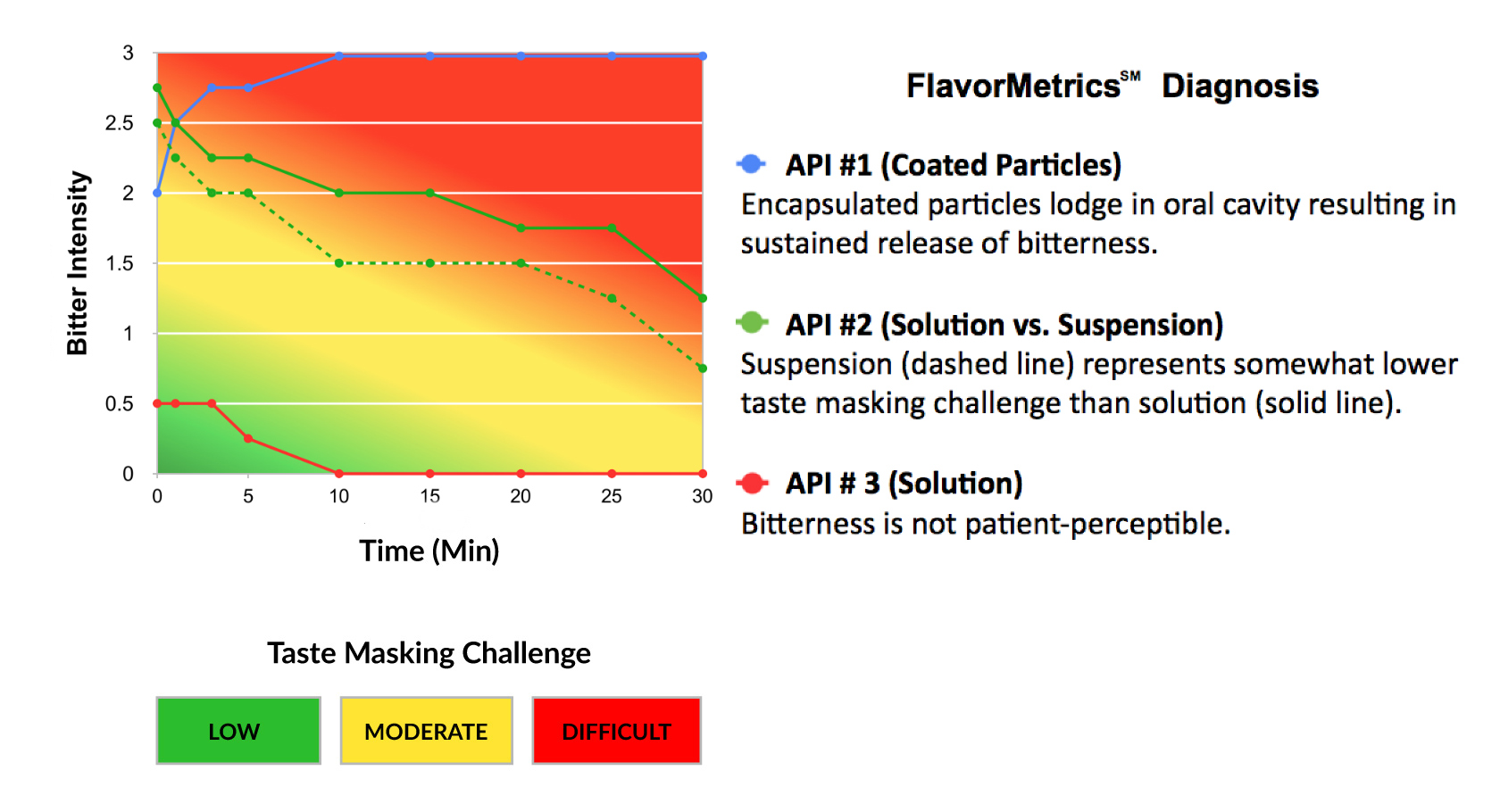
The taste masking challenge of an API is not a single value—it is a time-dependent sensory response driven by receptor activation and persistence.
Short, rapidly dissipating bitterness may be addressable with conventional approaches. In contrast, high-intensity or long-duration bitterness fundamentally constrains formulation strategy.
The FlavorMetrics℠ Bitterness Profile provides quantitative data to inform formulation development.
This step defines the feasible design space for taste masking.
Dose/Response Sensory Analysis
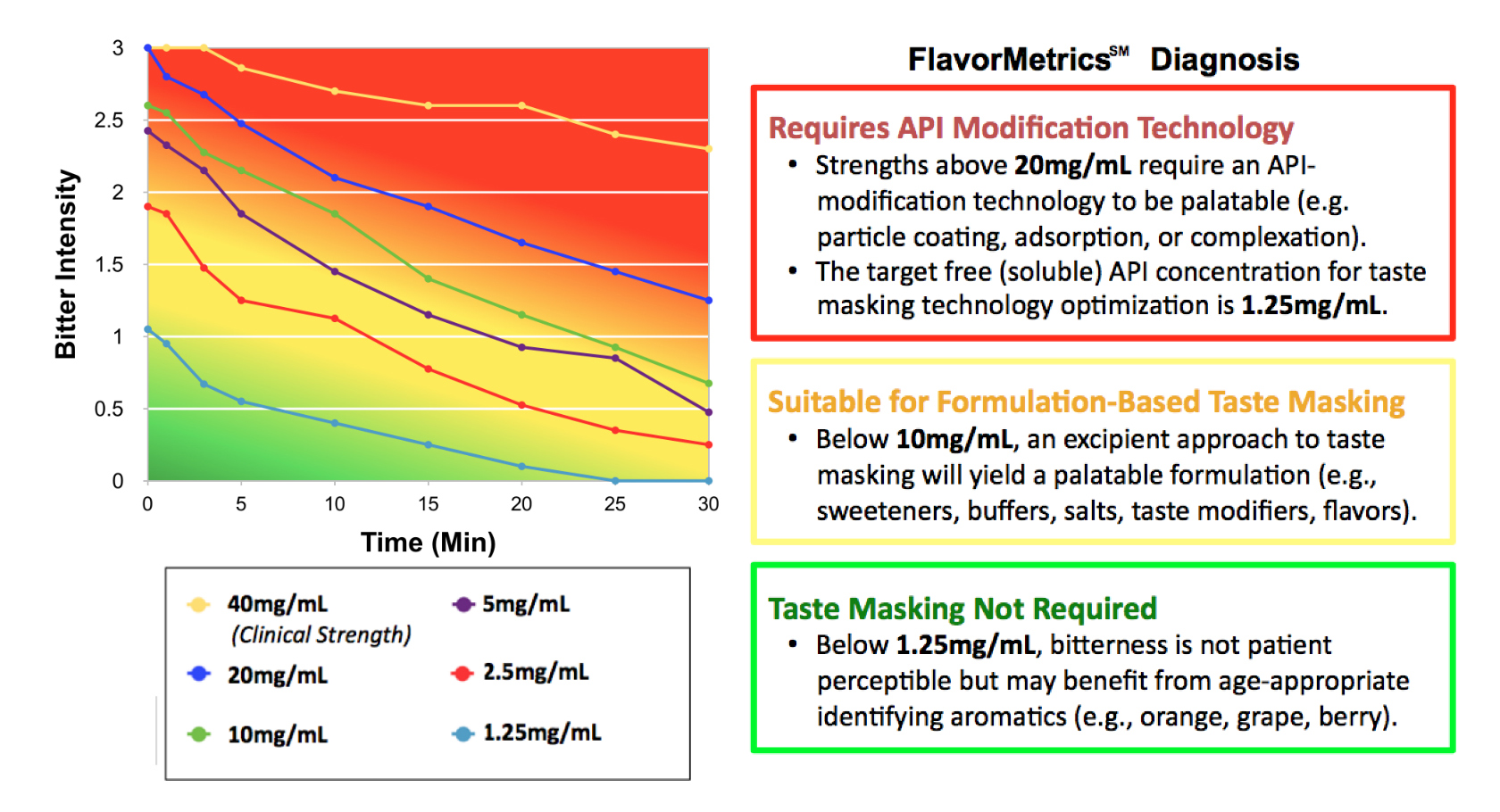
Taste masking is not simply about reducing bitterness—it is about understanding the constraints imposed by API solubility, dose, and release in the oral cavity.
Dose/response sensory analysis establishes these limits quantitatively:
Unlike conventional approaches that rely on arbitrary addition of flavor, this step defines what must be engineered out vs what can be masked.
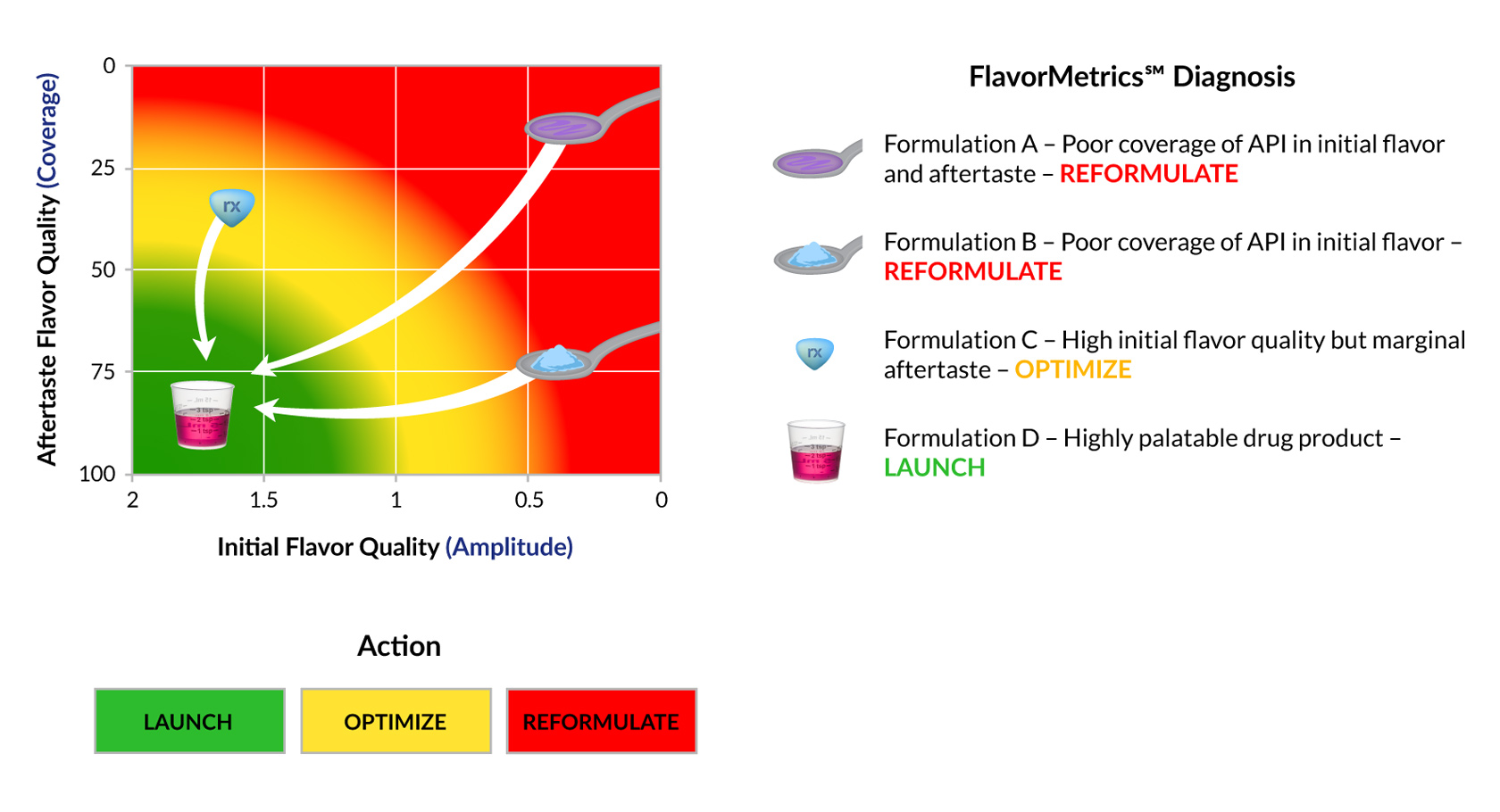
FlavorMetrics℠ Palatability Profile
Palatability is driven by more than initial taste. Products that are acceptable on first exposure but leave a persistent or unpleasant aftertaste often fail in real-world use.
The FlavorMetrics℠ Palatability Profile separates and quantifies the two key drivers of patient acceptance:
This empirical model enables:
By isolating these dimensions, formulation teams can systematically improve palatability rather than relying on overall liking scores, which often obscure critical deficiencies.
The data generated through comprehensive taste assessment directly informs formulation strategy—defining constraints, guiding technology selection, and enabling optimization against clear sensory targets.
The result is a development approach in which palatability is engineered with intent, reducing reformulation cycles, accelerating decision-making, and minimizing the risk of clinical or commercial failure due to poor taste.
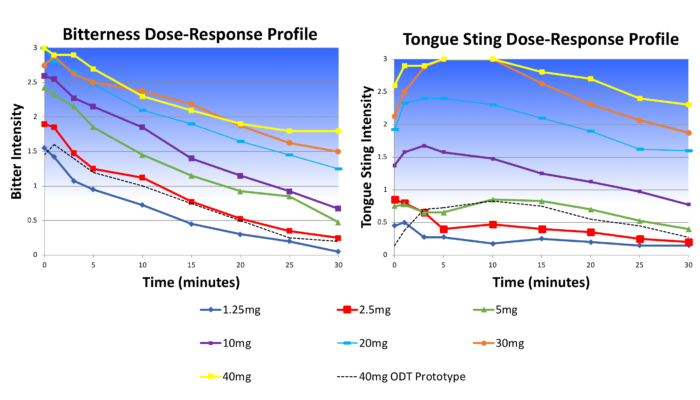
The client was developing an orally disintegrating tablet (ODT) form of an approved drug known to be extremely bitter. Senopsys conducted dose-response sensory analysis to establish the maximum free API concentration that could be effectively delivered in a palatable ODT and to evaluate the taste masking effectiveness of adsorption technology.

How good is your knowledge of taste masking? Learn the truths behind the science and what it means for your development program.
Yes. Trained adult sensory panels are widely used to identify and quantify aversive sensory attributes relevant to pediatric acceptance. In many cases, achieving levels below established recognition thresholds provides strong indication of improved palatability across patient populations.
Yes. Senopsys studies use controlled sensory methodologies and documented quality systems designed to support formulation decisions, technical reports, and regulatory submissions.
Trusted by 15 of the top 25 global pharma companies, Senopsys brings sensory science and formulation expertise to the most challenging taste masking problems.
Reach out to schedule a technical discussion with our experts.